The Future of Monitoring Lung Function in LAM

As all LAM patients are well aware of, serial lung function testing performed in a physician’s office is the current standard of care to assess the disease status for LAM patients. A full set of lung function tests comprises of the following:
1) Spirometry: measures the maximum amount of air you can exhale in the first one-second, known as forced expiratory volume in one-second (FEV1), and the total amount of air you can exhale, known as forced vital capacity (FVC).
2) Lung volumes: measure the total amount of air in your lungs at full breath, known as total lung capacity (TLC), and the volume remaining in your lungs after maximal exhalation, known as residual volume (RV).
3) Diffusion capacity: measures the ability of your lungs to transfer gas from the air into your blood, known as DLCO.
Of the above-mentioned components, spirometry, especially FEV1, is the most important element that we track over time in LAM patients. Although typically performed in a clinic/office-based setting, recent advances in technology have enabled the development of portable hand-held devices that can be used by patients to perform spirometry from the comfort of their home. The data obtained from these home-based spirometers can be easily transmitted via Bluetooth connection on a smartphone app or over the internet to central online portals, thus enabling easy access for both patients and their physicians.

Because of the day-to-day variability in spirometry, reliable estimates of disease progression typically require multiple measurements (3 or more) obtained over a prolonged period, usually 12 months or more. Home spirometry, by virtue of providing multiple measurements in a short time, can provide these estimates in a much more efficient manner, perhaps in as short a time frame as 3 months. For instance, office-based spirometry performed every three months will yield 4 FEV1 values in a year, whereas once weekly home spirometry measurements will provide 12 FEV1 values in 3 months. By providing access to more measurements, home spirometry can increase the precision of our assessment and shorten the time required to make meaningful conclusions regarding disease trajectory (Figure 1).
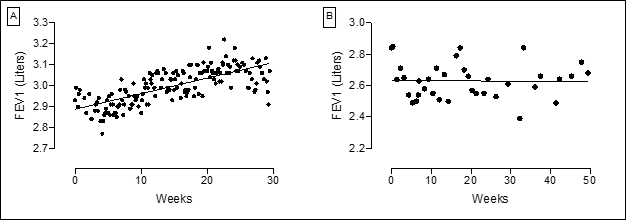
Figure 1: Home spirometry tracings from two LAM patients. Both tracings highlight the day-to-day variability in FEV1 thus stressing the importance of multiple measurements. The patient in Panel A performed home spirometry on an almost daily basis. As evidenced from the tracings, the improvement in lung function is quite clear even in a rather short monitoring time frame. The tracing in Panel B represents a LAM patient who performed home spirometry roughly once a week. Despite some variability in measurements, the number of values makes it possible to confidently assess the overall stability in this patient.
Home spirometry also offers other potential benefits for LAM patients. As access to routine office-based lung function testing remains limited during COVID-19, home spirometry provides a safe way to monitor lung function without exposing patients to undue risk. It is not hard to imagine a future where LAM patients could perform the majority of their routine lung function assessments by using home spirometry, limiting office-based testing to annual tests that include lung volumes and diffusion capacity measurements.
There are multiple aspects of home spirometry that need to be better studied prior to widespread use. There is a clear difference in the FEV1 values obtained via home spirometry compared with the office-based PFTs; the values obtained from home spirometry tend to be lower. As a result, the absolute values obtained by these modes of testing cannot be directly compared. However, the primary reason to obtain serial lung function testing in LAM is to determine trends rather than absolute values. Although this has not been well studied in LAM, we believe the longitudinal trends obtained from home spirometry will provide similar value as office-based PFTs.
How often should LAM patients perform home spirometry? Is home spirometry monitoring suitable for all LAM patients?
The correct answer to these questions is not yet known. We need to find a balance between the advantages of more frequent measurements and the burdens imposed by another regular disease-monitoring task for the patients. Potential applications of home spirometry include determining the pace of disease progression in newly diagnosed patients in order to guide decisions about starting treatment, assessing treatment response after starting sirolimus or modifying the dose, and monitoring patients enrolled in clinical trials. Finally, there are financial considerations affecting both patients and providers, as the cost of purchasing home spirometers is generally not covered by most insurance companies and physicians are not typically reimbursed for monitoring and interpreting home spirometry data.
The feasibility, reliability and overall performance of home spirometry in LAM is currently under investigation. Home spirometry was used as an exploratory end point in the recently concluded Resveratrol and Sirolimus in LAM Trial (RESULT, NCT03253913). Similarly, home spirometry is being studied as part of the ongoing Multicenter Interventional LAM Early Disease trial (MILED, NCT03150914). We are optimistic that in the near future home spirometry can empower LAM patients to safely monitor the status of their lung disease from the comfort of their home, provide investigators with an additional tool to assess treatment efficacy in clinical trials, shorten the time required to derive estimates of disease progression, and help answer other important questions such as the impact of menstrual cycle-related hormonal changes on lung function.

About Nishant Gupta, MD: Dr. Gupta is The LAM Foundation’s Scientific Director, and an Associate Professor in the Division of Pulmonary, Critical Care and Sleep Medicine at the University of Cincinnati, where he serves as the director of the interstitial and rare lung diseases program. Dr. Gupta’s clinical and research focus is on the field of rare lung diseases such as LAM, and his work is aimed at better defining the natural history, improving detection, and developing novel treatment modalities and monitoring strategies for LAM patients.